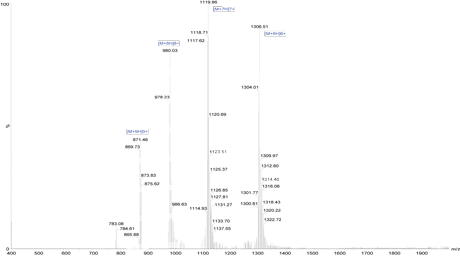
Catalytic active sites and most receptor binding sites for small ligands are found at domain and subunit interfaces. Long peptides are frequently synthesized to study the protein-receptor binding. Two active sites are indicated by the presence of the bound ligands (yellow). Both sites are at an interface between the two subunits of the protein.
We are asked frequently to synthesize relatively large peptides sized 100–200 residues, which is the size of a mean protein domain. We recently synthesized a 169-amino acid peptide. This demonstrates that the synthesis of peptides sized 100–200 amino acids is not beyond the capabilities of our current PeptideSyn technology. This long peptide was synthesized from intermediate fragments that were generated using stepwise SPPS, purified in their unprotected form, and ligated chemically with minimal protection.
A Simple Protocol to Refold Peptides or Small Proteins.
Obtaining peptides sized 100–200 amino acids using chemical synthesis is much faster and cheaper than cloning and overexpressing in Escherichia coli. In addition, the resulting peptide is always correct. Chemical synthesis can be used to incorporate non-genetically encoded structures, such as D-amino acids, into the protein in a completely regular fashion. Synthetic peptides eliminate problems such as poor or no expression, cloning errors, tags like FLAG or 6-His, or the mis-translation of non-preferred codons in prokaryotic hosts. Artificial amino acids that have isosteric side chains can be used to investigate the functional importance of specific residues.
Strategy for Synthesis-Solid Phase and hybrid technology
Pros
- Rapid synthesis of long peptides using an Fmoc/Boc strategy
- Short production cycles
- Automated and scalable procedure
- No isolation of intermediates
Cons
- Expensive raw materials, waste and operation
- Complex impurity profiles that include inserted, truncated, deleted, double-hit, modified, and unmodified amino acids
Case Study 1
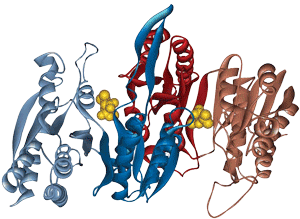
A 169-amino-acid peptide (MW 18716.41 Da, 83% purity) was synthesized successfully in 4 weeks. The success of the synthesis depends on the sequence: some 150 amino acid peptides can yield better results than those with 70 residues. Usually, Boc/Bzl protection is optimum for long or difficult sequences and base-sensitive peptides. Cleaving the peptide product from the resin requires strong acids such as TFMSA or HF. During the deprotection and cleavage from the resin, some reactive intermediates can react with vulnerable moieties such as Arg, Asp, or Glu in the peptide. LifeTein's PeptideSyn technology has a revolutionary effect on the synthesis of long peptides by eliminating these issues.
HPLC Purified Peptide
Case Study 2
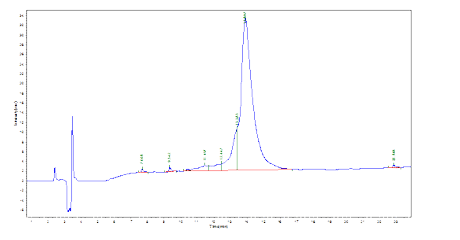
A client requested a very hydrophobic 68-amino-acid peptide (85% purity) with FITC modification at the N terminus. The peptide was successfully synthesized in 4 weeks.
HPLC Results
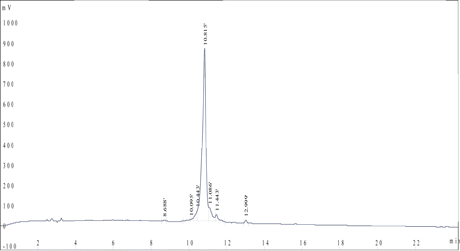
Mass Spectrometry Results